The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. 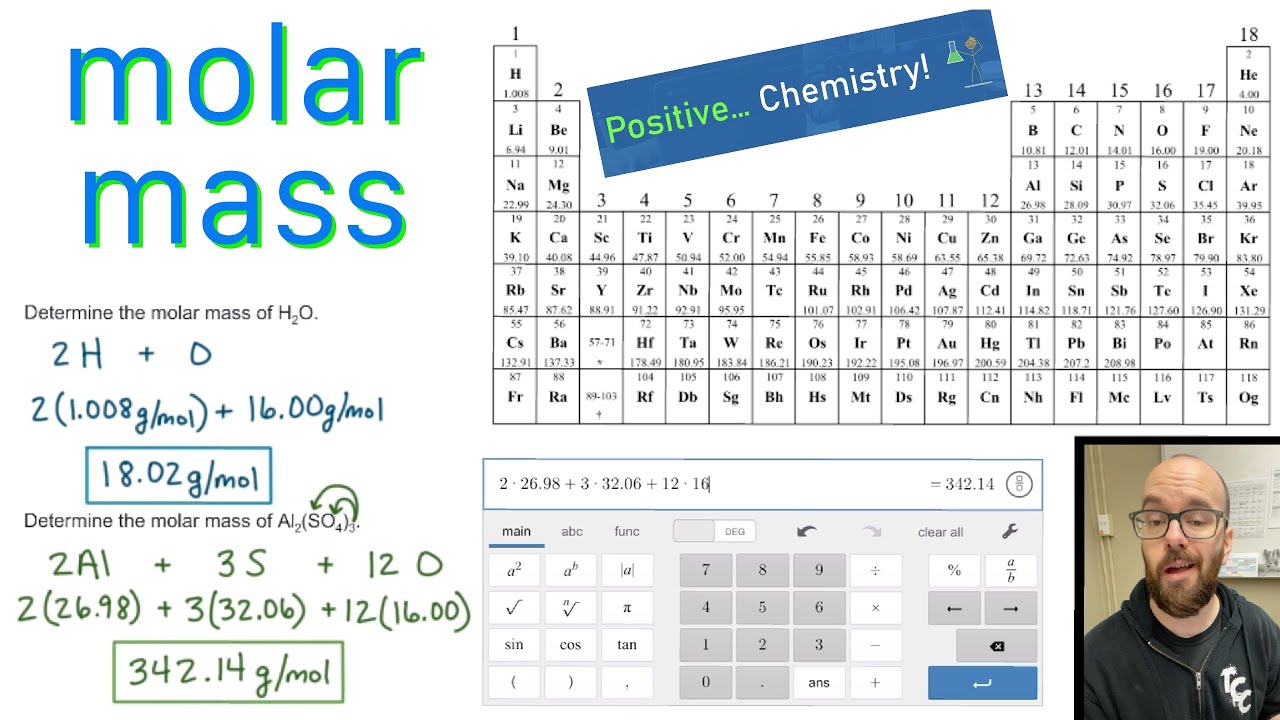
Lets calculate the molar mass of glucose. You will need to refer to a periodic table for proton values. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. How do we know the molar masses of individual atoms They are assigned to each element in this molar mass calculator or use the periodic table to read them. In this notation, the atomic number is not included. Atomic Weight is the molar mass of an element. Symbol-mass format for the above atom would be written as Cr-52. Rather inconveniently, the periodic table of elements reports the measured average mass of atoms in atomic mass units (amu), so a single hydrogen atom weighs. The unit for molar mass (note it is the mass of a mole) is grams/mole. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. What is the molar mass for lead(II) sulfate, PbSO4, Molar mass is the mass of one mole, in grams. Molar mass (2 x 1.008) + (2 x 15.999) Molar mass 34. masses of the individual elements on the periodic table. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Calculate the molar mass by multiplying the number of atoms of each element in the compound (its subscript) times the atomic mass of the element from the periodic table. Both the atomic number and mass are written to the left of the chemical symbol. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. The composition of any atom can be illustrated with a shorthand notation called A/Z format.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
